Therapeutic Potential of Cannabidiol in Alleviating Cognitive Decline and Hippocampal Damage in a Rat Model of Alzheimer's Disease
DOI:
https://doi.org/10.22452/mjs.vol44sp1.14Keywords:
Alzheimer’s disease, Cannabidiol, Cognitive deficits, Hippocampus, Neurotherapeutic effectsAbstract
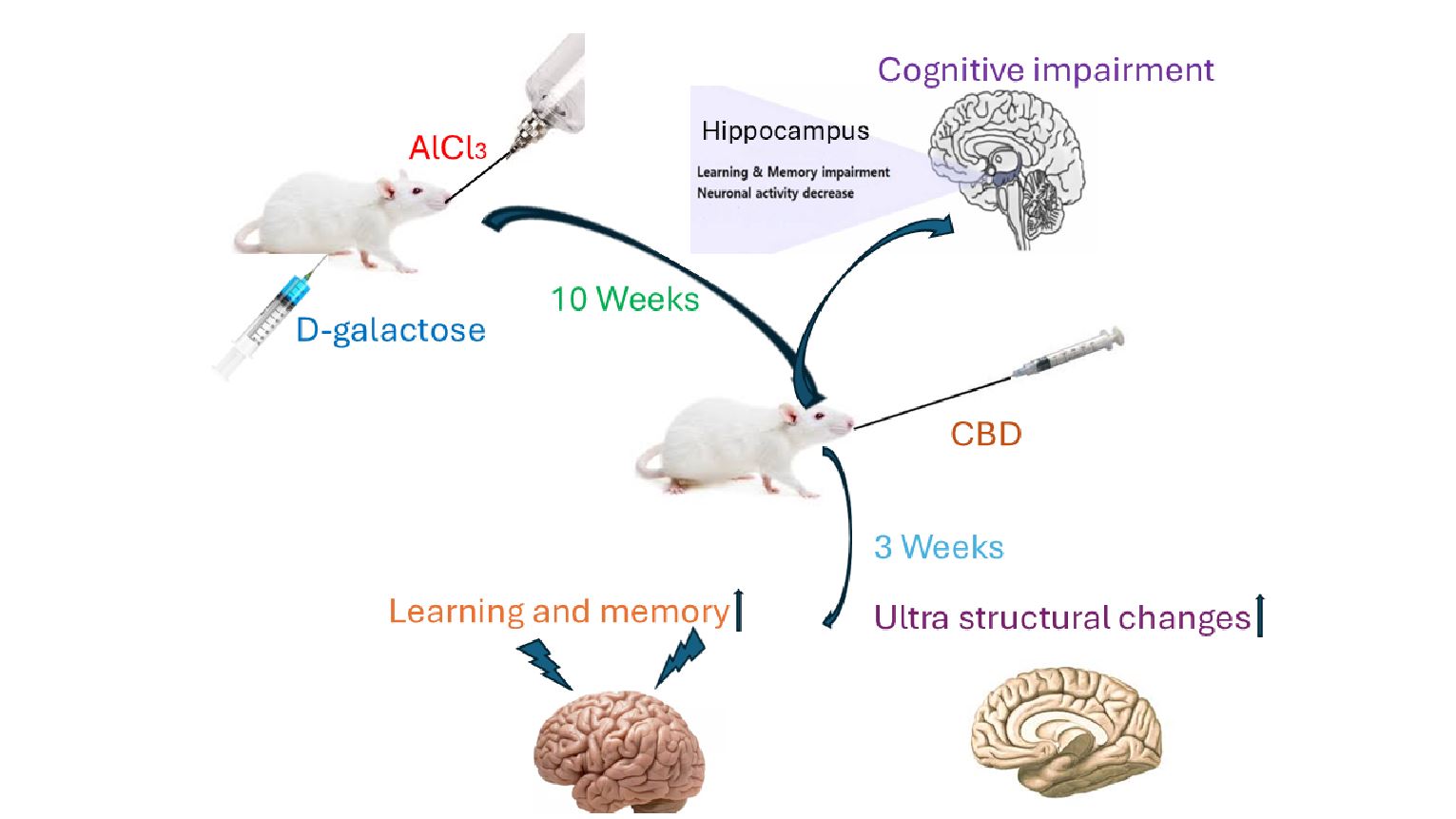
Alzheimer's disease (AD) is a common neurodegenerative disorder marked by progressive cognitive decline. Due to its effects on cognitive functioning and hippocampal integrity, the combined treatment of D-galactose (D-gal) and Aluminium chloride (AlCl3) in rats is a widely used model for producing AD-like symptoms. Previous studies demonstrated that Cannabidiol (CBD) exhibits neurotherapeutic effects. This study examines the efficacy of CBD in reducing cognitive deficits and brain ultrastructural damage induced by D-gal and AlCl3. Male Wistar rats were treated with D-gal (60 mg/kg body weight/day) and AlCl3 (200 mg/kg body weight/day) for 10 weeks to induce AD-like symptoms, followed by CBD administration at doses of 20, 40, and 80 mg/kg/day. Donepezil (1 mg/kg body weight/day) served as a positive control. Cognitive performance was evaluated using the modified elevated plus maze and T-maze spontaneous alternation tests. Ultrastructural changes in the hippocampus were examined using transmission electron microscopy. Rats exposed to D-gal and AlCl3 exhibited significant cognitive impairments, including deficits in spatial learning and memory, as well as hippocampal ultrastructural damage. The results indicated that D-gal and AlCl3 exposure produced notable cognitive deficits and structural alterations in the hippocampus. Administration of CBD at all doses significantly enhanced cognitive function and reduced pathological changes, providing protective effects comparable to donepezil. These findings support CBD's potential as a neurotherapeutic compound for mitigating cognitive decline and hippocampal damage associated with AD.
References
Abdelmeguid, N. E., Khalil, M. I. M., Elhabet, R., Sultan, A. S., & Salam, S. A. (2021). Combination of docosahexaenoic acid and Ginko biloba extract improves cognitive function and hippocampal tissue damages in a mouse model of Alzheimer’s disease. Journal of Chemical Neuroanatomy, 116, 101995. https://doi.org/10.1016/j.jchemneu.2021.101995
Bhunia, S., Kolishetti, N., Arias, A. Y., Vashist, A., & Nair, M. (2022). Cannabidiol for neurodegenerative disorders: A comprehensive review. Frontiers in Pharmacology, 13, 989717. https://doi.org/10.3389/fphar.2022.989717
Buccellato, F. R., D’anca, M., Fenoglio, C., Scarpini, E., Galimberti, D., & Barone, E. (2021). Role of oxidative damage in Alzheimer’s disease and neurodegeneration: From pathogenic mechanisms to biomarker discovery. Antioxidants, 10(9). https://doi.org/10.3390/antiox1009
Chiroma, S. M., Hidayat Baharuldin, M. T., Mat Taib, C. N., Amom, Z., Jagadeesan, S., Adenan, M. I., & Mohd Moklas, M. A. (2019). Protective effect of Centella asiatica against D-galactose and aluminium chloride induced rats: Behavioral and ultrastructural approaches. Biomedicine and Pharmacotherapy, 109, 853–864. https://doi.org/10.1016/j.biopha.2018.10.111
Chiroma, S. M., Mohd Moklas, M. A., Mat Taib, C. N., Baharuldin, M. T. H., & Amon, Z. (2018). d-Galactose and aluminium chloride induced rat model with cognitive impairments. Biomedicine & Pharmacotherapy, 103, 1602–1608. https://doi.org/10.1016/j.biopha.2018.04.152
Deacon, R. M. J., & Rawlins, J. N. P. (2006). T-maze alternation in the rodent. Nature Protocols, 1(1), 7–12. https://doi.org/10.1038/nprot.2006.2
Di Benedetto, G., Burgaletto, C., Bellanca, C. M., Munafò, A., Bernardini, R., & Cantarella, G. (2022). Role of microglia and astrocytes in Alzheimer’s disease: From neuroinflammation to Ca2+ homeostasis dysregulation. Cells, 11(17). https://doi.org/10.3390/cells11172728
d’Isa, R., Comi, G., & Leocani, L. (2021). Apparatus design and behavioural testing protocol for the evaluation of spatial working memory in mice through the spontaneous alternation T-maze. Scientific Reports, 11(1). https://doi.org/10.1038/s41598-021-00402-7
Feng, W., Qin, C., Cipolla, E., Lee, J. B., Zgair, A., Chu, Y., Ortori, C. A., Stocks, M. J., Constantinescu, C. S., Barrett, D. A., Fischer, P. M., & Gershkovich, P. (2021). Inclusion of medium-chain triglyceride in lipid-based formulation of cannabidiol facilitates micellar solubilization in vitro, but in vivo performance remains superior with pure sesame oil vehicle. Pharmaceutics, 13(9). https://doi.org/10.3390/pharmaceutics13091349
Flores-Cuadra, J. A., Madrid, A., Fernández, P. L., Pérez-Lao, A. R., Oviedo, D. C., Britton, G. B., & Carreira, M. B. (2021). Critical review of the Alzheimer’s disease non-transgenic models: Can they contribute to disease treatment? Journal of Alzheimer’s Disease, 82(s1), S227–S250. https://doi.org/10.3233/JAD-200870
Fordjour, E., Manful, C. F., Sey, A. A., Javed, R., Pham, T. H., Thomas, R., & Cheema, M. (2023). Cannabis: A multifaceted plant with endless potentials. Frontiers in Pharmacology, 14. https://doi.org/10.3389/fphar.2023.1200269
Ganguly, U., Kaur, U., Chakrabarti, S. S., Sharma, P., Agrawal, B. K., Saso, L., & Chakrabarti, S. (2021). Oxidative stress, neuroinflammation, and NADPH oxidase: Implications in the pathogenesis and treatment of Alzheimer’s disease. Oxidative Medicine and Cellular Longevity, 2021. https://doi.org/10.1155/2021/7086512
Hickey, J. P., Collins, A. E., Nelson, M. L., Chen, H., & Kalisch, B. E. (2024). Modulation of oxidative stress and neuroinflammation by cannabidiol (CBD): Promising targets for the treatment of Alzheimer’s disease. Current Issues in Molecular Biology, 46(5), 4379–4402. https://doi.org/10.3390/cimb46050266
Jagadeesan, S., Chiroma, S. M., Mohd Moklas, M. A., Hidayat Baharuldin, M. T., Mat Taib, C. N., Amom, Z., Vishnumukkala, T., Thomas, W., & Mahdi, O. (2022). Centella asiatica L. Urban protects against morphological aberrations induced by chronic unpredictable mild stress in rat’s hippocampus via attenuation of oxidative stress. Egyptian Journal of Basic and Applied Sciences, 9(1), 324–339. https://doi.org/10.1080/2314808X.2022.2091265
Jagadeesan, S., Gopalakrishna, P. K., Sura, S., Karikalan, B., Dandala, K. C. R., Ravindranadh, G., Moklas, M. A. M., & Vishnumukkala, T. (2024). Prevention of neuronal damage in brains of chronic stress induced male Wistar rats administering Centella asiatica (L) Urban. Journal of the Anatomical Society of India, 73(3), 204–213. https://doi.org/10.4103/jasi.jasi_80_24
Jagadeesan, S., Musa Chiroma, S., Baharuldin, M. T. H., Taib, C. N. M., Amom, Z., Adenan, M. I., & Moklas, M. A. M. (2019). Centella asiatica prevents chronic unpredictable mild stress-induced behavioral changes in rats. Biomedical Research and Therapy, 6(6), 3233–3243. https://doi.org/10.15419/bmrat.v6i6.550
Ju, Y., & Tam, K. (2022). Pathological mechanisms and therapeutic strategies for Alzheimer’s disease. Neural Regeneration Research, 17(3), 543–549. https://doi.org/10.4103/1673-5374.320970
Kamaruzzaman, M. A., Romli, M. H., Abas, R., Vidyadaran, S., Hidayat Baharuldin, M. T., Nasaruddin, M. L., Thirupathirao, V., Sura, S., Warsito, K., Mohd Nor, N. H., Azwaruddin, M. A., Alshawsh, M. A., & Mohd Moklas, M. A. (2023). Regulatory role of the endocannabinoid system on glial cells toward cognitive function in Alzheimer’s disease: A systematic review and meta-analysis of animal studies. Frontiers in Pharmacology, 14. https://doi.org/10.3389/fphar.2023.1053680
Khan, K., Emad, N. A., & Sultana, Y. (2024). Inducing agents for Alzheimer’s disease in animal models. Journal of Exploratory Research in Pharmacology, 000(000), 000–000. https://doi.org/10.14218/jerp.2023.00028
Lam, J., Katti, P., Biete, M., Mungai, M., Ashshareef, S., Neikirk, K., Lopez, E. G., Vue, Z., Christensen, T. A., Beasley, H. K., Rodman, T. A., Murray, S. A., Salisbury, J. L., Glancy, B., Shao, J., Pereira, R. O., Abel, E. D., & Hintonjr., A. (2021). A universal approach to analyzing transmission electron microscopy with imagej. Cells, 10(9). https://doi.org/10.3390/cells10092177
Luo, L., Yan, T., Yang, L., & Zhao, M. (2024). Aluminum chloride and D-galactose induced a zebrafish model of Alzheimer’s disease with cognitive deficits and aging. Computational and Structural Biotechnology Journal, 23, 2230–2239. https://doi.org/10.1016/j.csbj.2024.05.036
Mahdi, O., Chiroma, S. M., Baharuldin, M. T. H., Nor, N. H. M., Taib, C. N. M., Jagadeesan, S., Devi, S., & Moklas, M. A. M. (2021). Win55,212-2 attenuates cognitive impairments in AlCl3 + D-galactose-induced Alzheimer’s disease rats by enhancing neurogenesis and reversing oxidative stress. Biomedicines, 9(9), 1270. https://doi.org/10.3390/biomedicines9091270
Mutlu, O., Ulak, G., Celikyurt, I. K., Tanyeri, P., Akar, F. Y., & Erden, F. (2011). Effects of olanzapine and clozapine on memory acquisition, consolidation, and retrieval in mice using the elevated plus maze test. Neuroscience Letters, 501(3), 143–147. https://doi.org/10.1016/j.neulet.2011.07.004
Nabeshima, T., & Kameyama, T. (1990). Utility for the effects of Jiro Itoh 3, of an elevated plus-maze evaluation of memory in mice: Of nootropics, scopolamine, and electroconvulsive shock. Psychopharmacology, 101, Springer-Verlag.
Nascimento, A. B., Rodrigues, A. L. S., & Santos, A. R. S. (2020). The role of cannabinoids in the regulation of cognitive processes: A comprehensive review. Pharmacology & Therapeutics, 208, 107478. https://doi.org/10.1016/j.pharmthera.2019.107478
Schmitt, J. M., de Souza, L. P., & Silva, M. L. (2022). Neuroinflammation in Alzheimer's disease and potential therapeutic approaches. Neurochemistry International, 153, 104254. https://doi.org/10.1016/j.neuint.2021.104254
Sheppard, H. S., Perry, C. M., & Mitchell, S. J. (2023). The effects of cannabidiol on cognitive function and neurodegeneration: A systematic review and meta-analysis. Journal of Neurochemistry, 165(1), 1-18. https://doi.org/10.1111/jnc.15529
Solis, E., Zhang, J., & Reiner, D. J. (2021). Long-term memory improvement in Alzheimer’s disease models: Cannabidiol and the potential for neurogenesis. Journal of Alzheimer's Disease, 79(2), 305–319. https://doi.org/10.3233/JAD-200966
Wang, Y., & Liu, X. (2021). Cannabis-derived compounds in Alzheimer’s disease therapy: Mechanisms, evidence, and challenges. Frontiers in Neuroscience, 15, 668544. https://doi.org/10.3389/fnins.2021.668544
Zhang, R., & Wang, Y. (2023). Recent advances in the molecular mechanisms of neurodegeneration in Alzheimer's disease and their therapeutic implications. Neuropharmacology, 189, 108460. https://doi.org/10.1016/j.neuropharm.2021.108460
Zhang, J., Shao, J., & Yang, M. (2022). Targeting the endocannabinoid system in neurodegenerative diseases: A critical review of current findings. Frontiers in Pharmacology, 13, 904500. https://doi.org/10.3389/fphar.2022.904500
Zhuang, X., & Du, W. (2023). Alzheimer's disease and neuroinflammation: The role of cannabinoids and future therapeutic perspectives. Neuroscience Bulletin, 39(2), 215–226. https://doi.org/10.1007/s12264-023-00887-9
Downloads
Published
Issue
Section
License
Copyright (c) 2025 Malaysian Journal of Science

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Transfer of Copyrights
- In the event of publication of the manuscript entitled [INSERT MANUSCRIPT TITLE AND REF NO.] in the Malaysian Journal of Science, I hereby transfer copyrights of the manuscript title, abstract and contents to the Malaysian Journal of Science and the Faculty of Science, University of Malaya (as the publisher) for the full legal term of copyright and any renewals thereof throughout the world in any format, and any media for communication.
Conditions of Publication
- I hereby state that this manuscript to be published is an original work, unpublished in any form prior and I have obtained the necessary permission for the reproduction (or am the owner) of any images, illustrations, tables, charts, figures, maps, photographs and other visual materials of whom the copyrights is owned by a third party.
- This manuscript contains no statements that are contradictory to the relevant local and international laws or that infringes on the rights of others.
- I agree to indemnify the Malaysian Journal of Science and the Faculty of Science, University of Malaya (as the publisher) in the event of any claims that arise in regards to the above conditions and assume full liability on the published manuscript.
Reviewer’s Responsibilities
- Reviewers must treat the manuscripts received for reviewing process as confidential. It must not be shown or discussed with others without the authorization from the editor of MJS.
- Reviewers assigned must not have conflicts of interest with respect to the original work, the authors of the article or the research funding.
- Reviewers should judge or evaluate the manuscripts objective as possible. The feedback from the reviewers should be express clearly with supporting arguments.
- If the assigned reviewer considers themselves not able to complete the review of the manuscript, they must communicate with the editor, so that the manuscript could be sent to another suitable reviewer.
Copyright: Rights of the Author(s)
- Effective 2007, it will become the policy of the Malaysian Journal of Science (published by the Faculty of Science, University of Malaya) to obtain copyrights of all manuscripts published. This is to facilitate:
- Protection against copyright infringement of the manuscript through copyright breaches or piracy.
- Timely handling of reproduction requests from authorized third parties that are addressed directly to the Faculty of Science, University of Malaya.
- As the author, you may publish the fore-mentioned manuscript, whole or any part thereof, provided acknowledgement regarding copyright notice and reference to first publication in the Malaysian Journal of Science and Faculty of Science, University of Malaya (as the publishers) are given. You may produce copies of your manuscript, whole or any part thereof, for teaching purposes or to be provided, on individual basis, to fellow researchers.
- You may include the fore-mentioned manuscript, whole or any part thereof, electronically on a secure network at your affiliated institution, provided acknowledgement regarding copyright notice and reference to first publication in the Malaysian Journal of Science and Faculty of Science, University of Malaya (as the publishers) are given.
- You may include the fore-mentioned manuscript, whole or any part thereof, on the World Wide Web, provided acknowledgement regarding copyright notice and reference to first publication in the Malaysian Journal of Science and Faculty of Science, University of Malaya (as the publishers) are given.
- In the event that your manuscript, whole or any part thereof, has been requested to be reproduced, for any purpose or in any form approved by the Malaysian Journal of Science and Faculty of Science, University of Malaya (as the publishers), you will be informed. It is requested that any changes to your contact details (especially e-mail addresses) are made known.
Copyright: Role and responsibility of the Author(s)
- In the event of the manuscript to be published in the Malaysian Journal of Science contains materials copyrighted to others prior, it is the responsibility of current author(s) to obtain written permission from the copyright owner or owners.
- This written permission should be submitted with the proof-copy of the manuscript to be published in the Malaysian Journal of Science
Licensing Policy
Malaysian Journal of Science is an open-access journal that follows the Creative Commons Attribution-Non-commercial 4.0 International License (CC BY-NC 4.0)
CC BY – NC 4.0: Under this licence, the reusers to distribute, remix, alter, and build upon the content in any media or format for non-commercial purposes only, as long as proper acknowledgement is given to the authors of the original work. Please take the time to read the whole licence agreement (https://creativecommons.org/licenses/by-nc/4.0/legalcode ).





